AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic size trend11/28/2023 
Thus, it takes much more energy than just overcoming a larger ionic charge would suggest. Element \text B B has more electron-occupied shells than element \text A A. Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell). Elements \text A A & \text B B belong to the same group. The third IE, however, is over five times the previous one. The elements in groups (vertical columns). Overall, smaller atomic size means electrons are closer to the nucleus and pulled more strongly, leading to larger electronegativity. Describe and explain the observed trends in atomic size, ionization energy, and electron affinity of the elements. A higher effective nuclear charge causes greater attractions to the electrons, pulling the electron cloud closer to the nucleus which results in a smaller atomic radius. Atom size decreases across a period because the atomic number of an atom increases, meaning the positive charge of the nucleus pulls in electrons more strongly, leading to a stronger electronegativity. Across a period, effective nuclear charge increases as electron shielding remains constant. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. In general, atomic radius decreases across a period and increases down a group. The general trend is for ionization energy to decrease moving from top to bottom down a periodic table group. Following properties of elements show a very clear periodic trend in the periodic table. Moving left to right across a period, atomic radius decreases, so electrons are more attracted to the (closer) nucleus. Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals.(g) + e^− \nonumber \] The general trend is for ionization energy to increase moving from left to right across an element period. Consequently, beryllium is significantly smaller than lithium. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. 
This time, even though the number of protons increases by a lot, the electron valence shells do not. Again, this is due to the effective charge at the nucleus. Which of these elements has the highest first ionization energy oxygen. The radius increases as you move down a group (column) and decreases as you move from left to right across a period (row). This direction increases the size of the atom. Both atomic radius and ionic radius follow a trend on the periodic table. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. The other trend of atomic radius or atom size occurs as you move vertically down an element group. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. You will find separate sections below covering the trends in atomic radius, first ionisation energy, electronegativity, melting and boiling points, and density. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. This page explores the trends in some atomic and physical properties of the Group 1 elements - lithium, sodium, potassium, rubidium and caesium. /PeriodicTable-Trends-56a1310e5f9b58b7d0bcea8a-5c2d154e46e0fb0001f3647a.jpg) 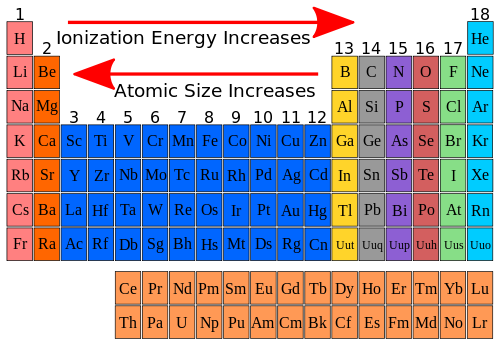
The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
